Learn More About BIS
Watch a short video that explains the technology
Give your patients peace of mind with the leading solution for point-of-care lymphedema assessment.
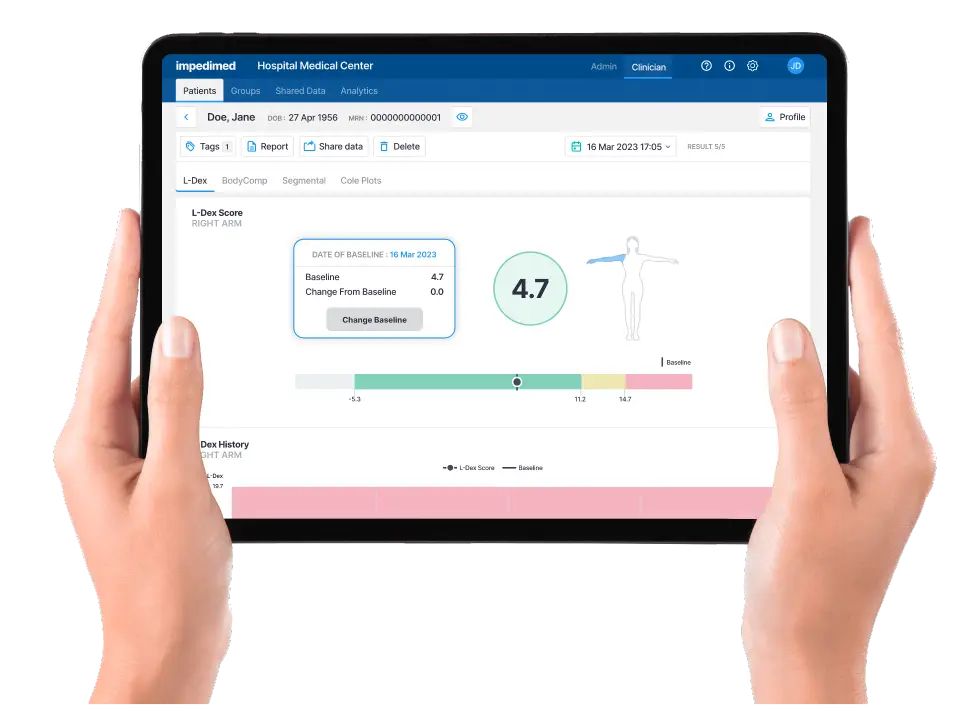
Lymphedema is rarely diagnosed until a patient experiences symptoms like pain, tightness,or swelling in their limb. However, with early diagnosis and intervention, the condition can be well managed1-3
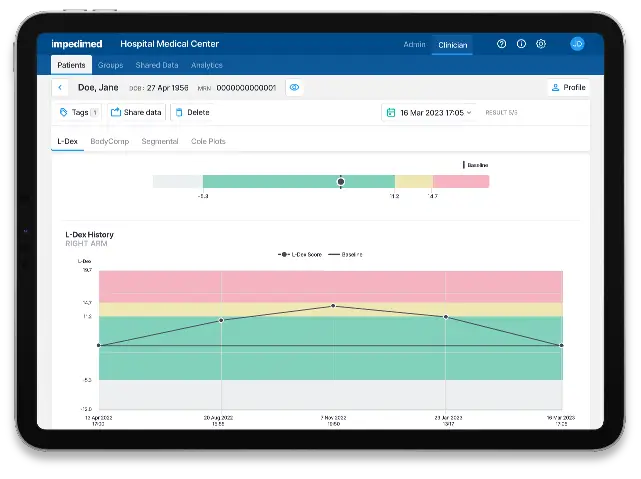

L-Dex analytics allows healthcare professionals to easily capture and visualize a patient’s pretreatment baseline L-Dex score and any follow up measurements.
An L-Dex increase of 6.5 or more from baseline is an indication that lymphedema is developing, and intervention is needed.
Consistent monitoring with L-Dex Analytics can give patients peace of mind, offering clear outputs of lymphedema-related fluid changes in the limbs.
![]()