1,200
BREAST CANCER PATIENTS
“[L-Dex] screening should be a standard approach for prospective breast cancer-related lymphedema surveillance.” –
The PREVENT Trial

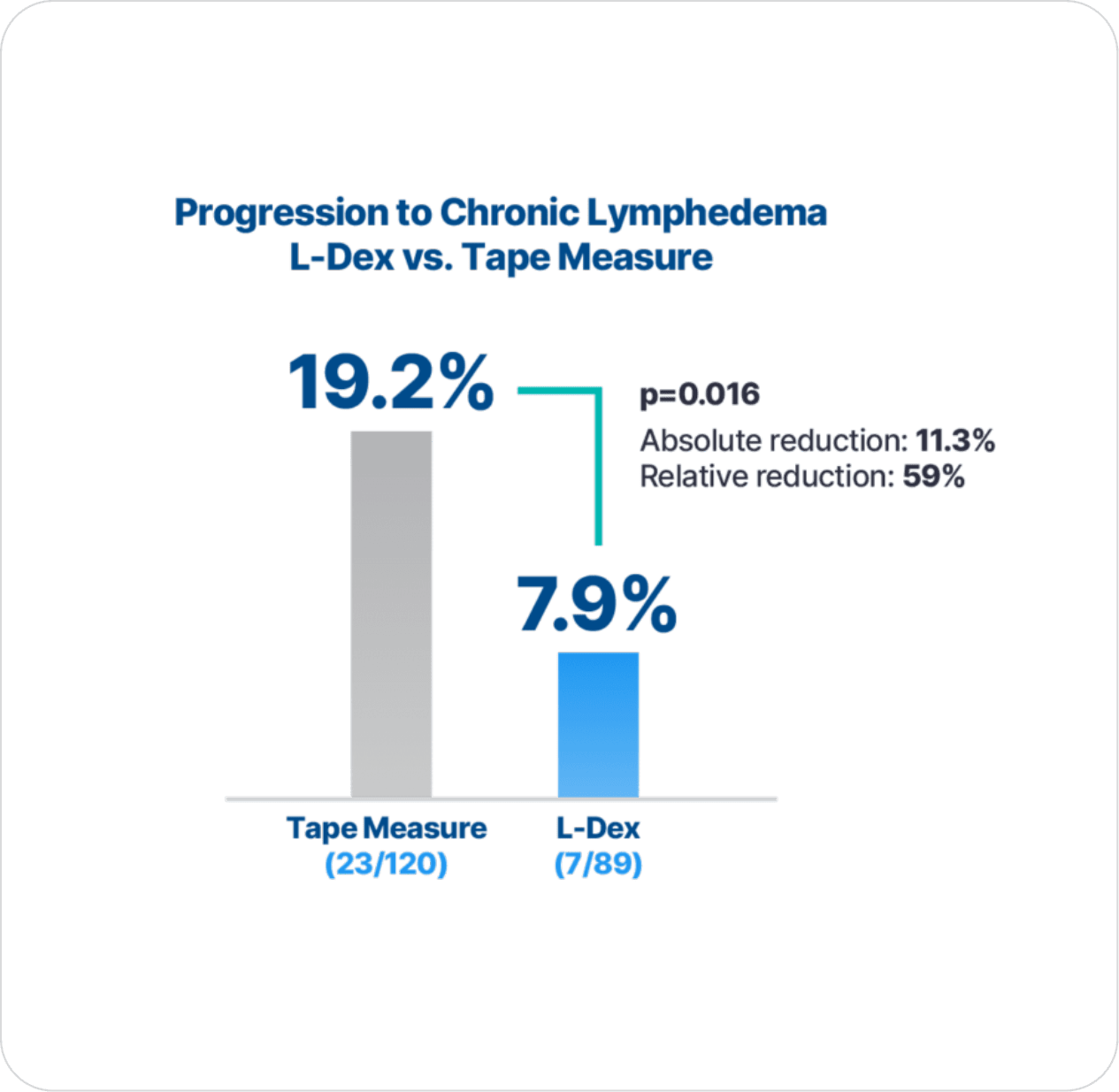
92% of patients with early detection using L-Dex® and intervention did not progress to chronic lymphedema.
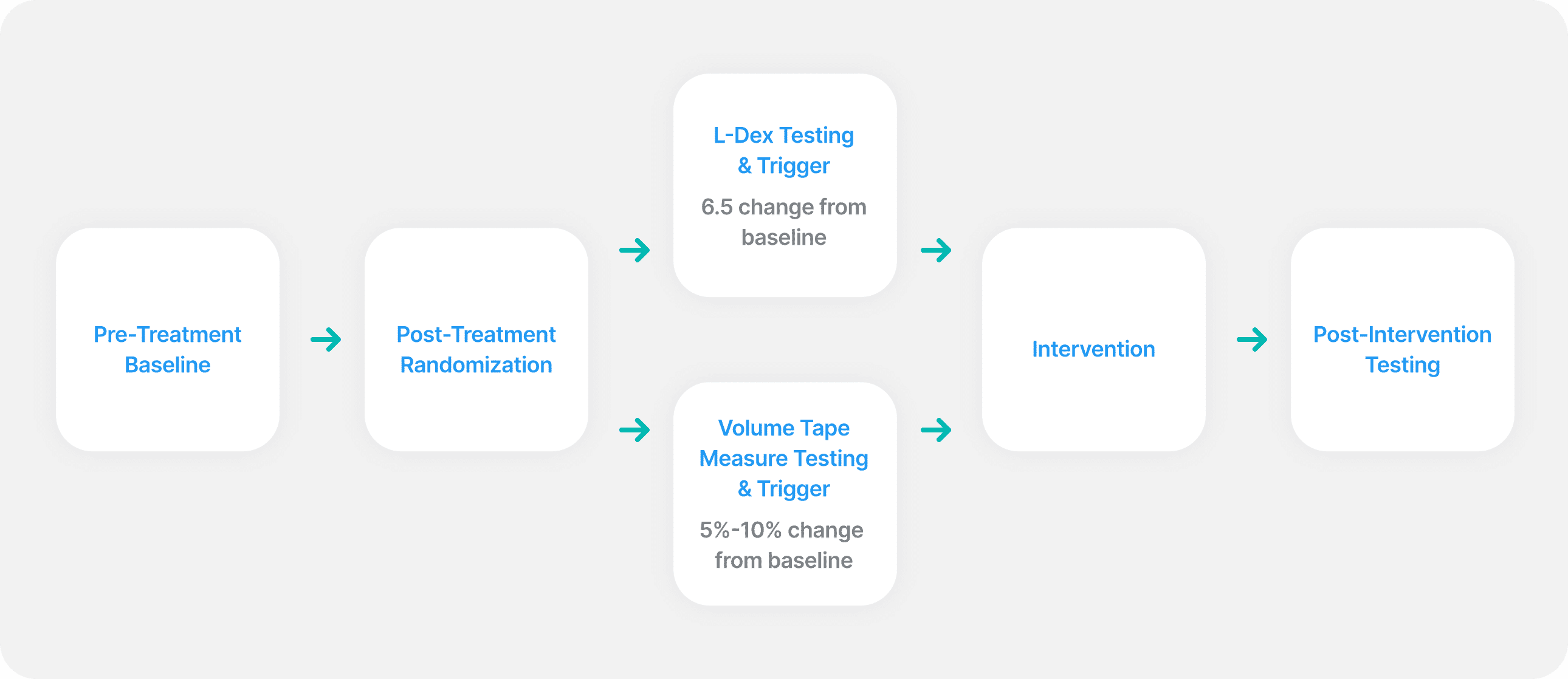
Newly diagnosed breast cancer patients with planned surgery received informed consent and baseline measurements using L-Dex and tape measure. Following surgery, patients were randomized to lymphedema follow-up using either L-Dex or tape measure.
Measurements were taken at 3, 6, 12, 15, 18, 21, 24, 30, 36 months with the 15 and 21 month visits at investigator discretion.
An L-Dex increase of 6.5 or more from baseline was a trigger for intervention in the L-Dex group. A tape measure volume increase between 5% and 10% from baseline was a trigger for intervention in the tape measure group.
Intervention was a 23-32 mmHG compression sleeve and gauntlet worn for 4 weeks, 12 hours a day.
Progression to complete decongestive physiotherapy (CDP) was the same for both groups: a tape measure volume increase of 10% or more from baseline.
with early detection using L-Dex and intervention versus using tape measure.
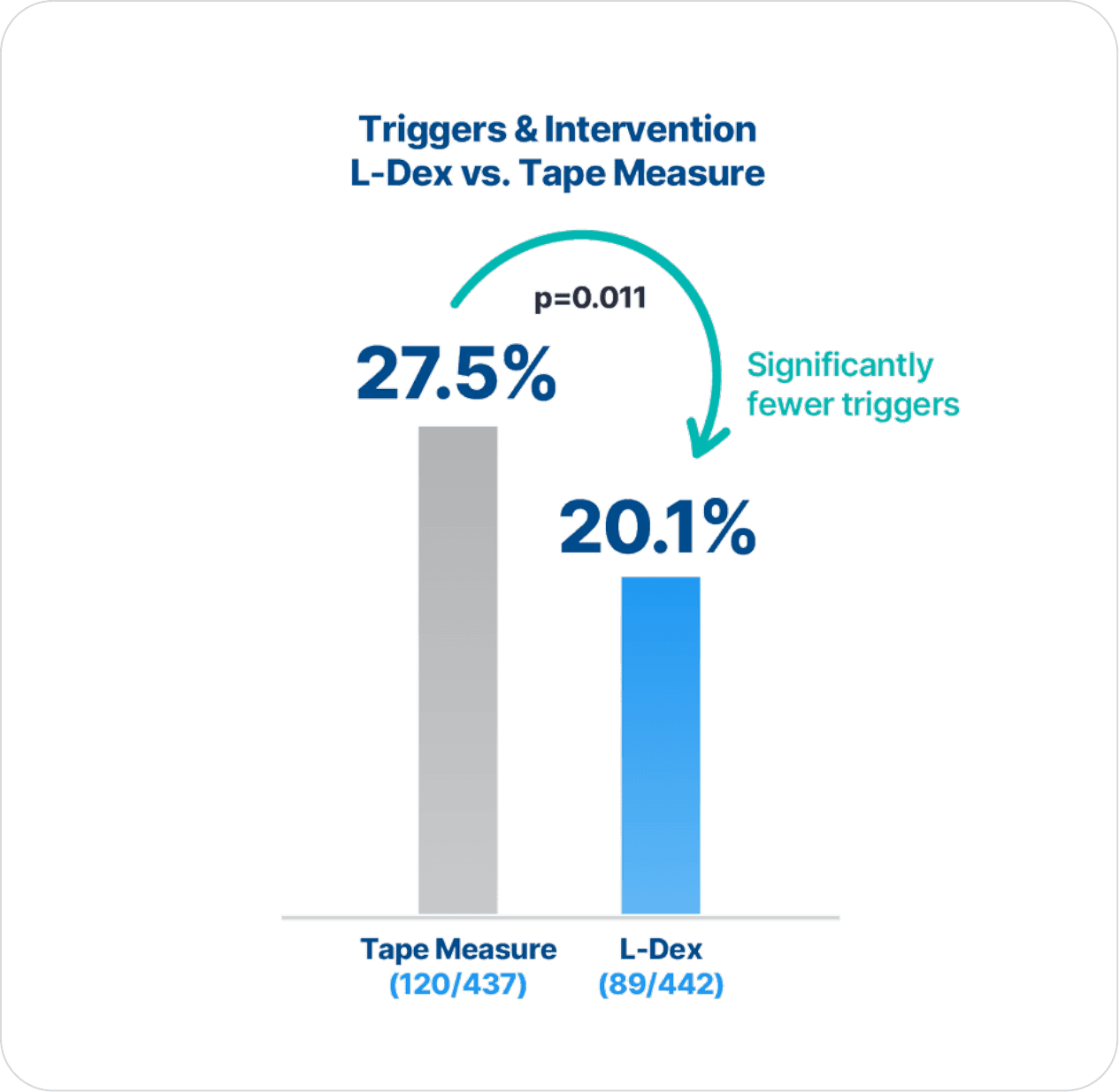
“[L-Dex] is more specific for lymphedema detection than tape measure.”
“[L-Dex], as compared to tape measure, provides a more precise identification of patients likely to benefit from an early compression intervention.”
Thank you to the patients and to the PREVENT investigators. Your contributions are advancing the science of lymphedema care so that cancer patients and survivors can access early detection and intervention of lymphedema.










Contact us to learn more about implementing L-Dex testing.
*Required