Educate & Inform
Your doctor, nurse, or therapist may not be aware of the latest technologies for early detection of lymphedema. Print this handout and take it to your next visit. Ask them what they think.
You can take control of your survivorship with simple, early lymphedema detection – before it becomes chronic.

Breast cancer-related lymphedema is a devastating side effect of breast cancer treatment.
1 in 5 breast cancer survivors will be impacted by arm lymphedema resulting from their cancer treatment1:
If lymphedema is left untreated:
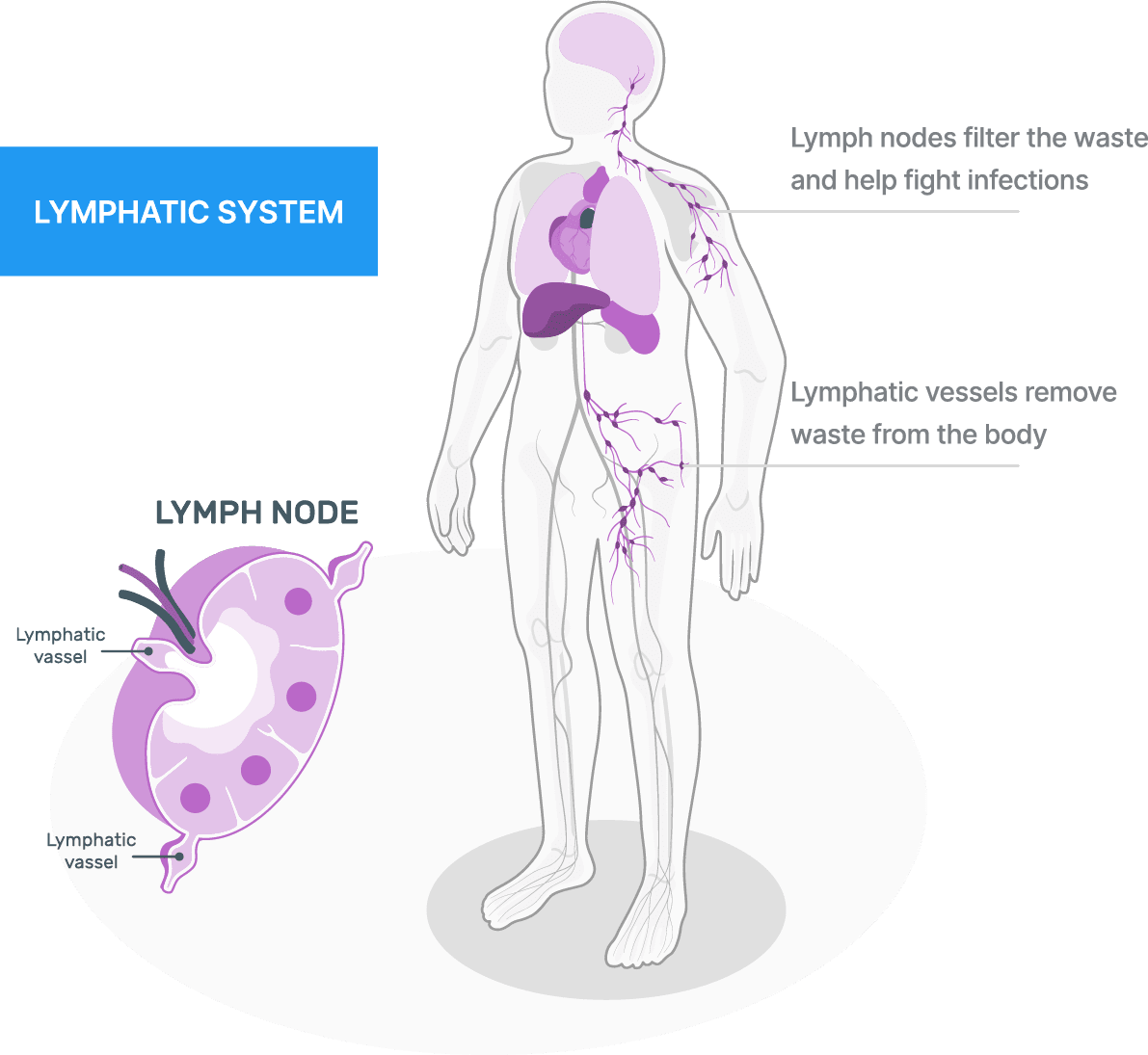
The lymphatic system runs like a super highway in your body. Certain cancer treatments damage and block the lymphatic system. Lymphedema occurs when fluid builds up due to a block in the lymphatic system.
You can take control of your survivorship with simple, early lymphedema detection – before it becomes chronic.

The opportunity to prevent lymphedema is before noticeable swelling exists.
You can take control of your survivorship with simple, early lymphedema detection – before it becomes chronic.

The SOZO® device is used to measure your L-Dex® score. It looks like a scale, but it has places for you to put your hands and feet. You will not feel the SOZO test and it only takes 30 seconds to complete. Your cancer care team uses your L-Dex score to help determine whether you have early lymphedema. If early lymphedema is identified, they will provide you with a personal treatment plan.

You can take control of your survivorship with simple, early lymphedema detection – before it becomes chronic.

Find a place near you to get your L-Dex score tested.
Your cancer care team is there to support you through diagnosis, treatment, and survivorship.
Use these tools to talk to your cancer care team about getting tested with your L-Dex score.
L-Dex testing is offered by healthcare professionals. To find a location near you, visit our Find a Provider page.
If your cancer care team does not offer L-Dex testing, they may still offer lymphedema management services. Not sure how to start a conversation about lymphedema? Print these questions and ask them during your next visit. Your doctor, nurse, or therapist may not be aware of the latest technologies for early detection of lymphedema. Print this handout and take it to your next visit. Ask them what they think.
Individual insurance carriers have different policies about covering the cost of an L-Dex test. To learn more, contact your insurance carrier or ask your healthcare provider.
Breast cancer-related lymphedema is a devastating side effect of breast cancer treatment. If left untreated, lymphedema can lead to painful swelling, infection, and hospitalization. It can require lifelong management and interfere with activities like work and caring for your family. Studies show that if lymphedema is detected early, then it can be treated and prevented from getting worse. The opportunity to prevent lymphedema, is before you experience noticeable swelling. Testing with the L-Dex score is shown to detect lymphedema early.
You should get tested with your L-Dex score as soon as you know you will be treated for cancer. Testing should continue during and after treatment. This way, your cancer care team can determine whether you have early lymphedema and should be treated.
If you already have symptoms of lymphedema, contact your cancer care team. Getting treatment as soon as possible will provide you with the best possible outcomes.
Individual insurance carriers have different policies about covering the cost of lymphedema treatment. To learn more, contact your insurance carrier or ask your healthcare provider.
Your cancer care team can refer you to the best place to get compression garments to treat lymphedema. Organizations like LIVE Today Foundation offer free-of-charge compression garments to those who need them. Visit our Community page or LIVE Today Foundation to learn more.