NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Survivorship
The NCCN Guidelines® are consistent about the importance of early detection of lymphedema and use of an objective tool, such as bioimpedance spectroscopy.3
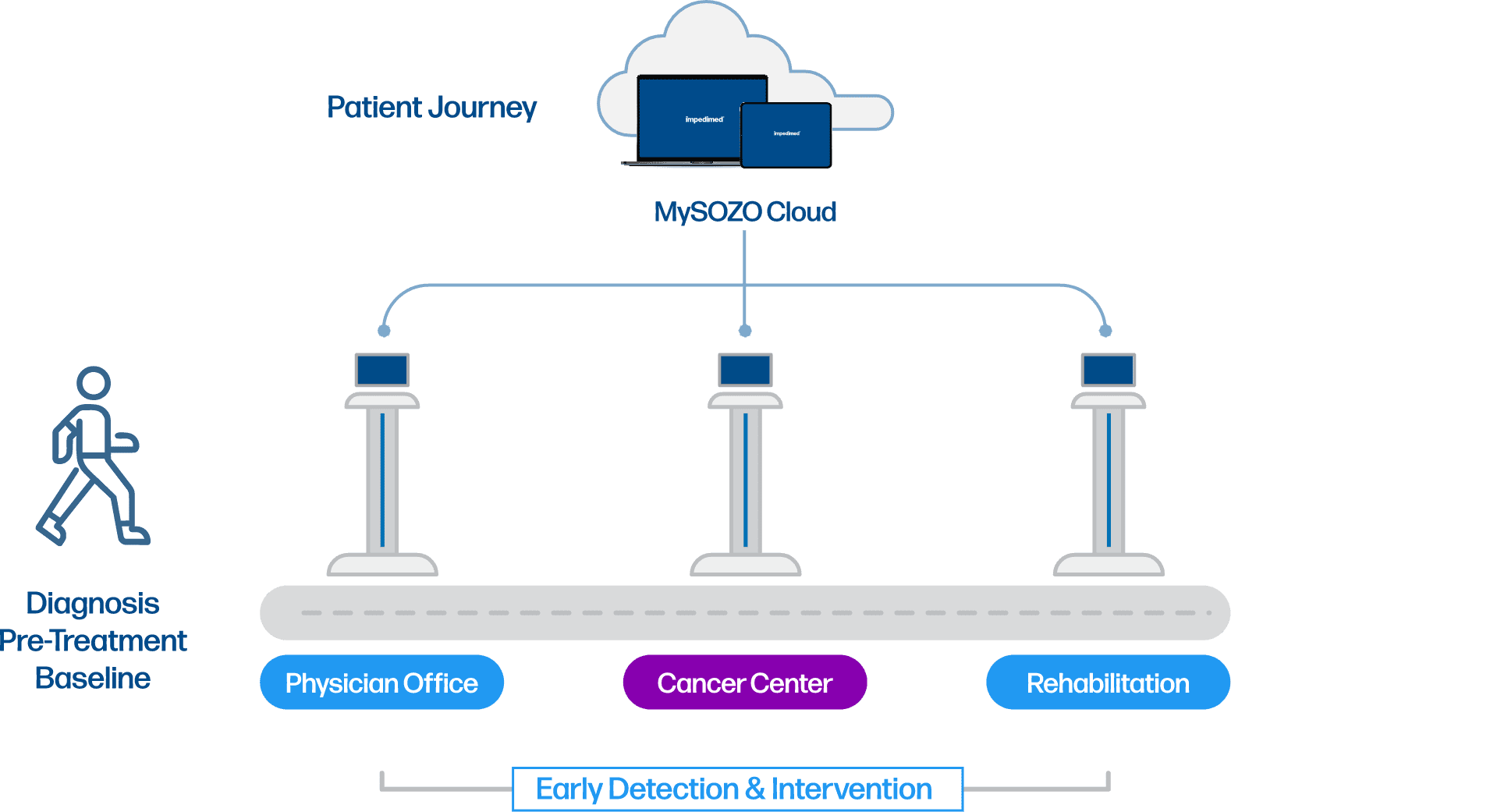
The SOZO® Digital Health Platform provides clarity across the cancer journey starting at diagnosis through survivorship, by providing real-time insights into how the body is changing, enabling earlier intervention by identifying trends before symptoms arise from problems like lymphedema and loss of muscle mass.

Up to 58% of patients with cancer are at risk for limb lymphedema, and this risk can climb to over 80% with breast cancer patients depending on their cancer treatment.1
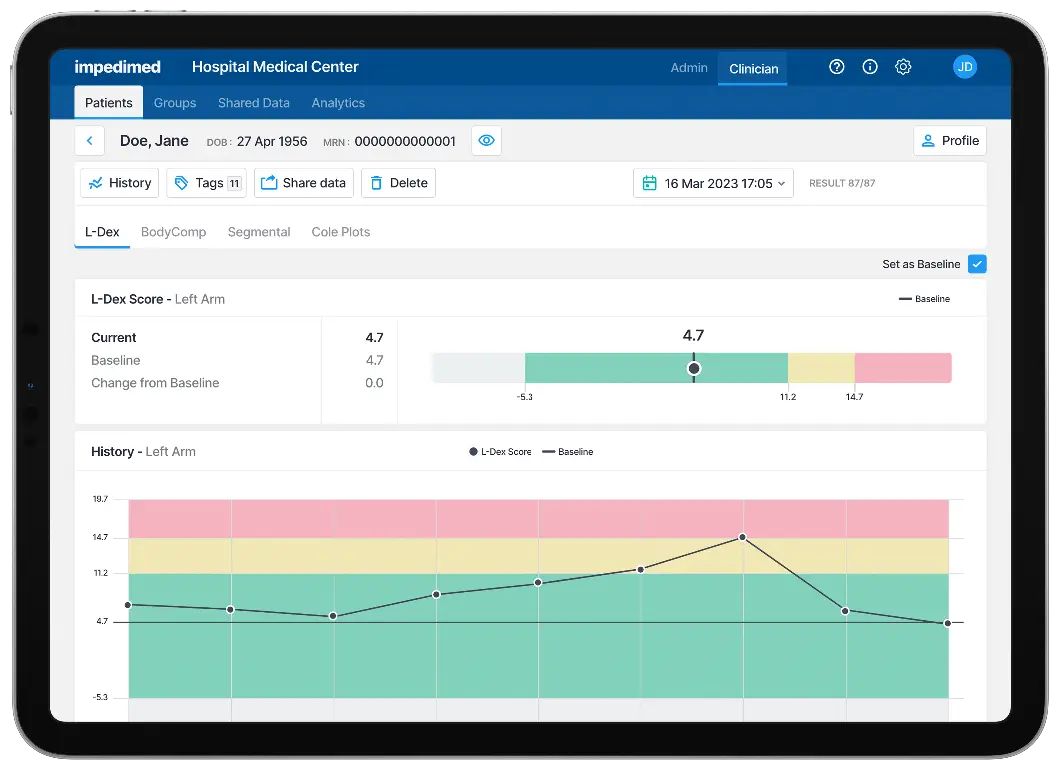
SOZO® Digital Health Platform with L-Dex is the only FDA-cleared bioimpedance spectroscopy (BIS) solution for the clinical assessment of lymphedema. By detecting lymphedema early at a subclinical level, clinicians can intervene and potentially reverse this condition.
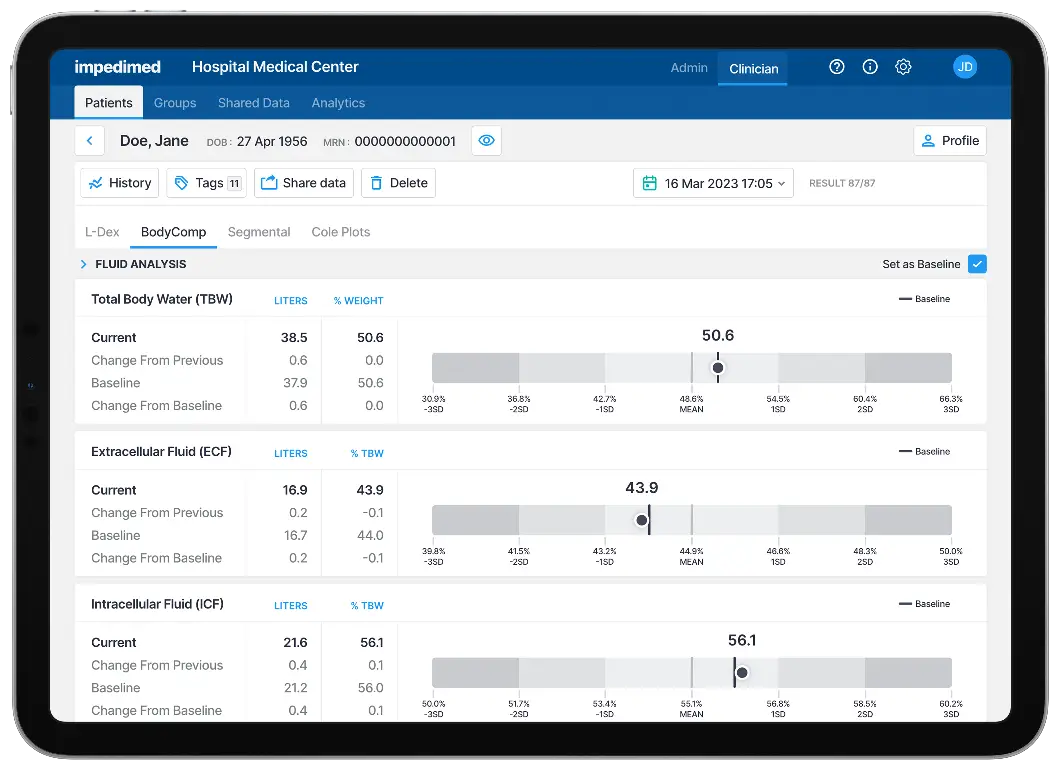
It’s important to look beyond BMI to determine predictors of cancer recurrence, mortality, and quality of life in survivorship. Muscle and fat mass are what matters.2 BodyComp delivers these insights, supporting patient conversations around:
In PREVENT, the largest randomized controlled trial (RCT) for lymphedema prevention in breast cancer patients, 92% of patients with early detection using BIS and intervention did not progress to chronic lymphedema.

Using our SOZO Digital Health Platform with L-Dex and BodyComp Analysis, ImpediMed partners with you to implement a prospective surveillance model to help detect lymphedema in cancer patients at the early stage, so that it can be treated and stopped from progressing to a lifelong condition, as well as monitoring key health indicators to support improved outcomes. Because care is tailored to how the body is genuinely responding — not just to reported symptoms — providers can personalize treatment, and patients are empowered by staying engaged and informed.